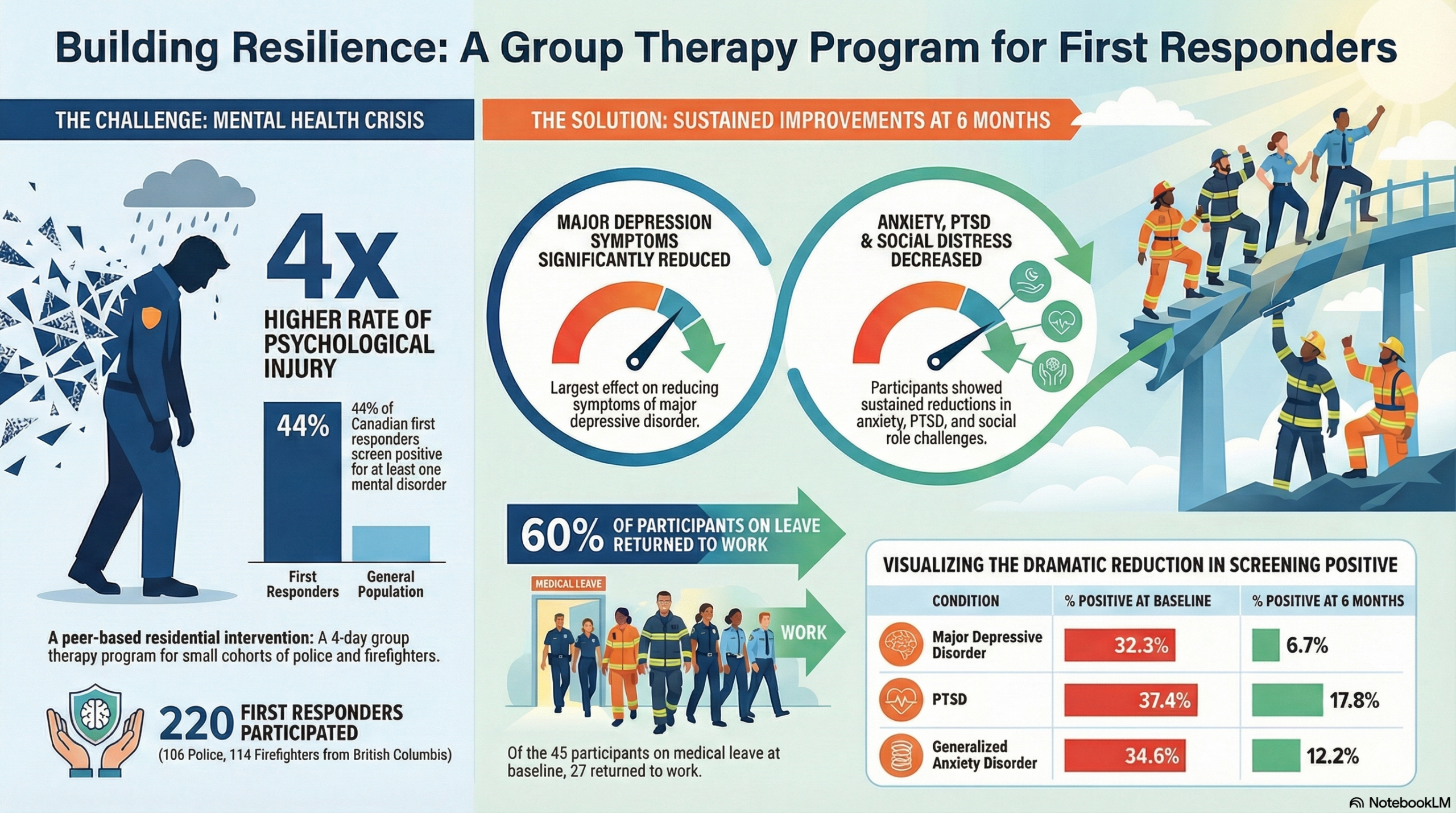
This blog summarizes the results of a recent study published in Comprehensive Psychiatry evaluating the First Responder Resiliency Program (FRRP). 220
Study recommends moving beyond opioid sparing
BC learns lessons from opioid prescribing portraits, new primary care study says. What is the study about? This study was part of a rigorous
20 years qualitative research interviewing
I first interviewed a person with lived experience of substance use twenty years ago. Today, I wanted to pause and reflect on the key points in this
Sending prescribing feedback won’t curb opioid use disorders
REDONNA was a single-blinded, two-arm clinical trial of a prescribing Portrait for eligible family physicians in British Columbia, Canada. It examined
Shifting Focus, Accelerating Innovative Vending Machines
Shifting focus on accelerating innovative solutions and clarifying roles can help reduce unregulated drug deaths. Bardwell and colleagues(1)
How can people with opioid use disorder get better virtual care?
Virtual care is the new normal around the globe. The emergence of COVID-19 introduced a dual public health emergency in British Columbia. The province
Can online training uplift opioid sparing strategies?
Opioid analgesic prescribing has been in trouble for decades in North America. Things are changing, though. Still, there’s room to adjust care,
Can intentional substance use reduce illicit opioid use?
Have you ever intentionally used an alternate substance to control or reduce your use of illicit (non-prescribed) opioids? Self-managed use is one
Hospital based opioid agonist treatment
Researchers recently found that many people with co-occurring mental health and substance use disorders are admitted to inpatient psychiatric units.