BC learns lessons from opioid prescribing portraits, new primary care study says. What is the study about? This study was part of a rigorous
20 years qualitative research interviewing
I first interviewed a person with lived experience of substance use twenty years ago. Today, I wanted to pause and reflect on the key points in this
Sending prescribing feedback won’t curb opioid use disorders
REDONNA was a single-blinded, two-arm clinical trial of a prescribing Portrait for eligible family physicians in British Columbia, Canada. It examined
Connecting professionals to promote interdisciplinary trauma-focused therapy
Connecting professionals can promote interdisciplinary trauma-focused therapy and return-to-work support for clients with work-related PTSD. This blog
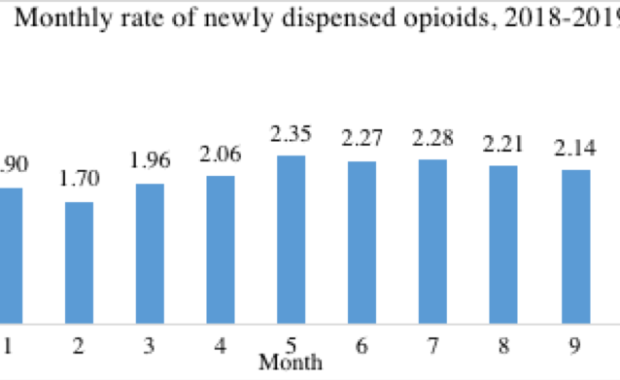
Newly started primary care opioid prescriptions plateaued in 2018-2019
Our new study published in the Canadian Journal of Addiction found plateauing rates of new starts of opioid analgesics in British Columbia in
Mentoring helps physicians publish more peer-reviewed papers, study says
Physician scientists help advance the science of addiction medicine, yet prior studies have not looked at better ways of increasing publication
Individualized prescribing portraits to reduce inappropriate initiation of opioid analgesics to opioid naïve patients in primary care, REDONNA trial
REDONNA is a mixed-methods randomized study testing the effectiveness of individualized prescribing Portraits to reduce inappropriate initiation of
Does this opioid naïve patient really need potent opioids? Think twice
Think Twice, exercise caution and generally avoid making assumptions about patient risk of subsequent prescription opioid use disorder, write authors
Can stimulants treat stimulant use disorder?
This ‘systematic overview of reviews’ assembled the evidence for stimulant use disorder treatments from the systematic reviews of literature.